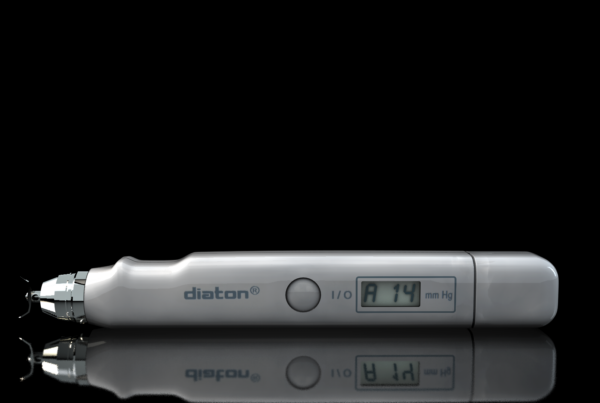
Medical Device Sales Increase at BiCOM Due to Diaton Tonometer – Glaucoma Eye IOP Test Through the Eyelid
BiCOM Inc. is one of the few US medical device companies that has been experiencing staggering growth within last 4 years. Since obtaining FDA clearance for its unique glaucoma IOP eye test through the Eyelid – Diaton Tonometer, the company has increased direct sales force and now covers every US state. Internationally, Diaton Tonometer is currently available in over 60 countries through various medical device distributors. CEO of BiCOM, Roman Iospa predicts for healthcare sector and medical diagnostic technology sales to increase.
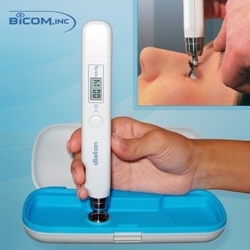
BiCOM has been in business for 18 years and has managed to build a solid brand around this new and unique transpalpebral tonometry technology which allows to determine intraocular pressure (IOP) through the EYELID http://www.tonometerdiaton.com . Traspalpebral (Diaton) Tonometry clientele consists of optometrists and ophthalmologists, military, nursing homes, hospitals, emergency rooms, general medical practitioners and family physicians, as well as some referred patients requiring day or night eye pressure monitoring.
In an interview with Roman Iospa, Chief Executive of BiCOM said, – “We plan to grow Diaton tonometer sales through our broader expansion internationally and continue to gain greater penetration of the market nationally, mainly due to greater awareness and an invaluable addition of new talent with new contacts and years of experience from market leading companies such as Alcon (NYSE:ACL), Abbott Laboratories (NYSE:ABT), Medtronic (NYSE:MDT), CIBA Vision, Novartis (NYSE:NVS), Kimberly-Clark (NYSE:KMB), Johnson & Johnson (NYSE:JNJ), GlaxoSmithKline (NYSE:GSK), Bausch & Lomb, Advanced Medical Optics, Pfizer (NYSE:PFE) and Allergan (NYSE:AGN) just to name a few…”
“Our transpalpebral (through the eyelid) Diaton tonometry is unique, effective and is irreplaceable in various situations where any other corneal tonometry methods (Goldmann, Tonopen, Air puff) simply can not be used”. Transpalpebral approach is ideal for patients with corneal concerns like conjunctivitis, cornea pathology, keratitis, keratocone, keratoplastik, edema, erosion, high degree of ametropy or astigmatism, keratoprosthesis, or acquiring IOP following laser refractive (LASIK, LASEK, PRK) and other corneal surgeries, “Diaton tonometer has a number of indisputable advantages of measuring through the upper lid and over the sclera (no contact with cornea) without need of anesthetic drops, making the IOP measuring process handy and economical for the doctor and comfortable for the patient,” Mr. Iospa said.
An increase in sales can be attributed to a proper business plan execution and advanced marketing techniques by BiCOM management team and a new set of clinical trials presented by reputable and well-known doctors at various industry events.
In an interview with Ocular Surgery News at the Association for Research in Vision and Ophthalmology (ARVO) meeting, Dr. Mark Latina said IOP measurements with the Diaton tonometer are equivalent to Goldmann applanation tonometry. The Diaton device offers a safe, noncontact method of measuring pressure via the eyelid, he said.
"It works," said Dr. Mark Latina, a glaucoma specialist and the inventor of Selective Laser Trabeculoplasty (SLT), "It gives you a reasonable estimation of the pressure without having to anesthetize the cornea through a transpalpebral approach."
Results of clinical studies, video presentations and articles are available on the company's website: http://www.TonometerDiaton.com
We have asked Mr. Iospa if President Barack Obama’s healthcare plan will have an effect on the future Diaton tonometer sales forecasts, to which Mr. Iospa has replied, “In short, if it will have an effect, only a positive one. Evidence suggests that preventive medical care through early diagnosis will save money in the long run and Obama’s health care plan aims to do just that. We have a diagnostic device on the market and with early detection and treatment, glaucoma can in most cases be controlled and vision preserved. Per Obama’s plan more people will have medical insurance and will need to be screened, which in turn will create more demand for our diagnostic product.”
BiCOM, a privately held company has increased its outlook for 2009 due to its plans to expand the product reach domestically and internationally. A growing need for glaucoma screenings in the aging population globally, combined with the unique transpalpebral method, allows BiCOM to position itself strongly within the growing eyecare diagnostic and medical devices industry.
BiCOM management also confirmed their expansion plans to introduce two additional medical products this year, a result of a number of earlier investments BiCOM made into medical innovation; details and amounts were not disclosed.
About BiCOM Inc.
BiCOM, with its 18 years of tradition and global clientele, has proven to be the enterprise of the highest level of professionalism, integrity and financial solvency. A unique team of engineers, medical, legal and business experts makes BiCOM Inc. the right place for global talent to find support and guidance. BiCOM Inc. sees its mission in bringing to market innovative products developed and manufactured worldwide.
BiCOM represents and supports Diaton tonometer in over 60 countries through its international medical equipment and ophthalmic devices distributors and growing.