This is Commodity Code 42183018 in UNSPSC Codes, the Commodity Name is Ophthalmic tonometers or accessories, more detail follows below:
Diaton Tonometer (Product Number N477) delivered complete with 2 year warranty, a carry case, calibration & training plate, battery, and training DVD in 5 different languages, manuals, quick starter guide, plus phone based training support. International and US contact info at 1-877-342-8667
Support via email: Contact@TonometerDiaton.com / Online: www.TonometerDiaton.com
Thank you for choosing Diaton tonometer – Unique tonometry through the eyelid & sclera!
If you have Any questions – We would be happy to hear from you. Please find contact options below.
Please NOTE: This item will not be shipped until payment is completed or Purchase Order PO is received.

If you have already completed your payment, a confirmation email is on its way with your order number and shipping details.
To complete your order, a payment can be made via our secure Online Payment Form or by phone at 1-877-DIATONS (877-342-8667)
** Purchase Orders can be emailed to contact@tonometerdiaton.com or faxed to (888) 260-0606.
If you require an Official QUOTE, please contact or obtain it via Online Payment Form on the main site.
Diaton – One Tonometer – Many Applications!
Please review numerous clinical studies showing Diaton in use in various interesting cases, such as Boston KPro, eyes that are affected by corneal pathologies which make corneal tonometry inaccurate, and often impossible – corneal injury, corneal edema, keratoconus, atypical corneal thickness, corneal rigidity, keratitis, cornea dimness, post keratoplastics, keratoprosthesis, post LASIK, high degree of ametropy, astigmatism, just to name a few.
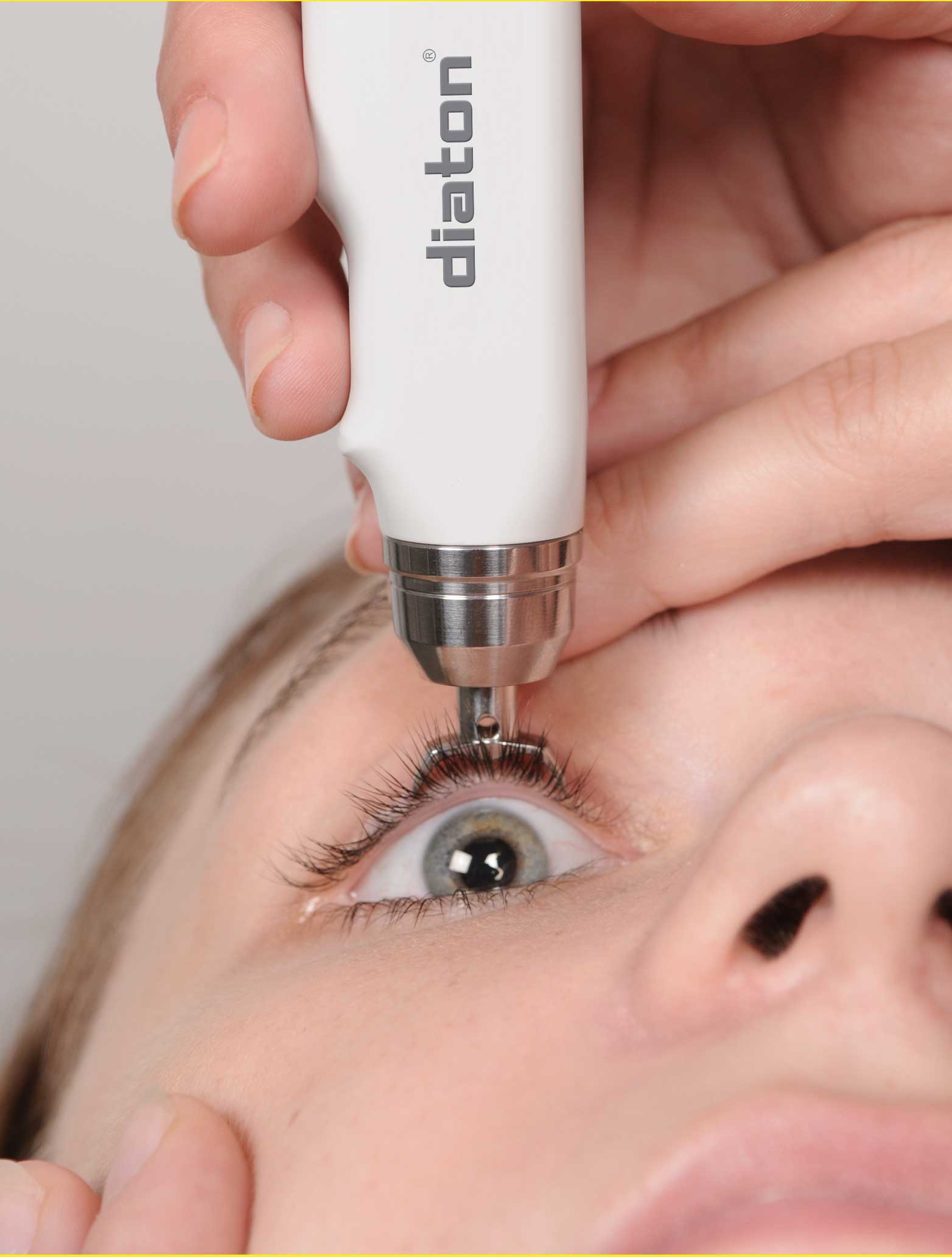
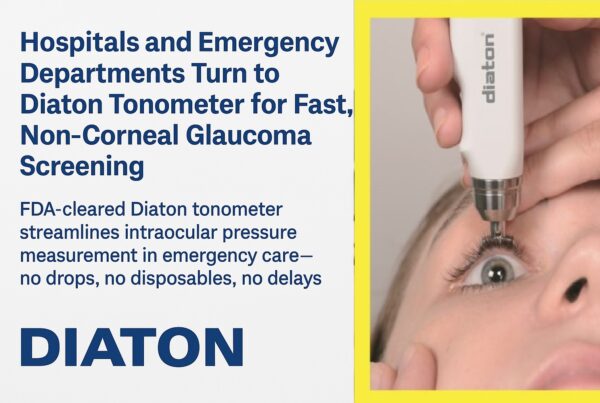
Because of it’s unique transpalpebral and transscleral method of measuring intraocular pressure (IOP), Diaton is a clinically necessary and a preferred choice for 1000’s of professionals. It’s quick, painless and accurate!
—— Major Benefits of Diaton Tonometry ——
– No contact with the cornea (only upper eyelid and sclera)
– No need for anesthesia drops
– No risk of infecting or scratching cornea (safer than other methods)
– No consumables (no need to purchase replacement tips/covers. Latex-free)
– No sterilization (just alcohol swab is used to wipe off the tip)
– No pachymetry needed (more accurate – independent of CCT and corneal biomechanics)
– No daily calibration needed (ready-to-use and saves time)
– Handheld, portable, pen-like
– Easy to use (can be used by any trained personnel, Step-by-step Videos Included)
For more information about Diaton tonometer visit www.TonometerDiaton.com
Easy-to-follow User Guide, Manuals & Training Videos at www.tonometerdiaton.com/guides-training/
Please feel free to contact us – we are always glad to answer any additional questions or process your order via phone.
Thank you for your business!
Team Diaton
DevelopAll Inc.
Toll Free: 1-877-DIATONS (1-877-342-8667)
Email: Contact@TonometerDiaton.com
Fax: 1-888-260-0606
Representing and Supporting Diaton in over 60 Countries….
Product Classification
FDA
Medical Devices
DatabasesDevice tonometer, ac-powered
Regulation Description Tonometer and accessories.
Regulation Medical Specialty Ophthalmic
Review Panel Ophthalmic
Product Code HKX
Premarket Review Ophthalmic, Anesthesia, Respiratory, ENT and Dental Devices (OHT1)
Ophthalmic Devices (DHT1A)
Submission Type 510(k)
Regulation Number 886.1930
Device Class 2
Total Product Life Cycle (TPLC) TPLC Product Code Report
GMP Exempt? No
Summary Malfunction
Reporting Eligible
Recognized Consensus Standard
10-96 ANSI Z80.10-2014
American National Standard for Ophthalmics - Ophthalmic Instruments - TonometersImplanted Device? No
Life-Sustain/Support Device? No
Third Party Review Not Third Party Eligible