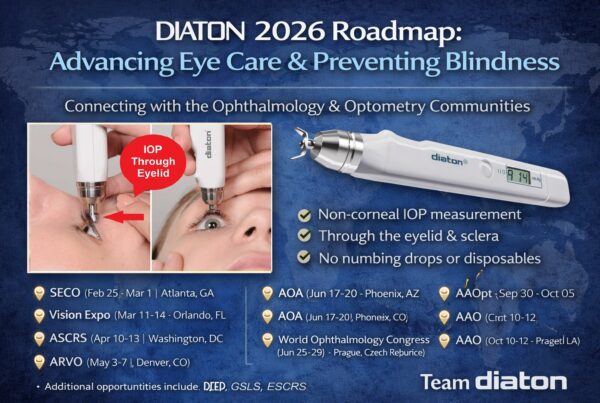
Transpalpebral technique
Pen-like tonometer designed to be patient-friendly
Noncontact device measures IOP quickly, shows results similar to ‘gold standard’ in studies
By J e n n i f e r A. W e b b
Reviewed by John Hope, MD, and Mark Latina, MD
Long Beach, NY-A handheld tonometer that measures IOP through the eyelid and over the sclera is proving to be helpful for ophthalmologists faced with patients who are apprehensive about seeing an instrument approaching their eyes or who have a corneal abnormality, according to several ophthalmologists who have studied it.
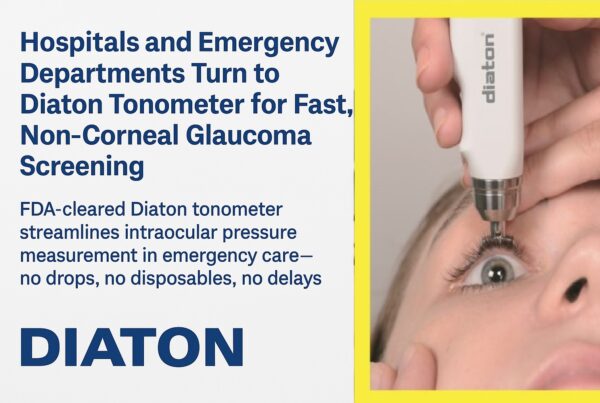
The tonometer (Diaton, DevelopAll Inc., New York) is a pen like instrument that measures IOP within seconds without the need for anesthesia or sterilization. Approved by the FDA, the instrument has been the subject of numerous clinical trials, where it has been found to be comparable with the gold standard the Goldmann applanation tonometer.
According to Roman Iospa, the company’s chief executive officer, the device is available in more than 50 countries, and more than 25,000 units are on the market. Additional accessories are not required.

Convenience for all
The device appeals to ophthalmologists who see pediatric patients and those with patients who have corneal edema or erosions or keratoprosthesis, he said. Because the device is used on the upper eyelid and out of the patients’ field of vision as they recline and look at a 45° angle, patients do not tend to blink or squeeze their eyes shut before the reading, which can skew the IOP measurement.
“It is convenient for the doctor and pain less for the patient,” Iospa said. “There is really no discomfort, especially for the patient who might be slightly anxious.”
Mark Latina, MD, and Tarek Shazly, MD, members of the Department of Ophthalmology, Massachusetts Eye & Ear Infirmary, Boston, and Emil William Chynn, MD, an ophthalmologist in private practice at Park Avenue Laser in New York, performed a study of the tonometer to compare IOP measurements taken with the device with those from the Goldmann applanation tonometer in normal and glaucomatous eyes.
The study examined 66 eyes of 33 consecutive subjects, 46 eyes having glaucoma and 20 eyes without. Goldmann tonometry was performed by one of the authors, whereas the proprietary device measurements were taken by another author in a masked fashion.
In both the normal and glaucoma groups, 15.15 % of the device measurements were exactly the same as those obtained with the Goldmann tonometer. The device underestimated the IOP compared with the Goldmann in 37.87% of eyes, and it overestimated the IOP in 43.93% of eyes.
The proprietary tonometer, however, correlated within 3 mm Hg of Goldmann in 83.3% of eyes and “may be a clinically useful screening device for measuring IOP,” the authors concluded.
“At the end of the study, we reached the conclusion that the transpalpebral technique is a very promising method for tonometry, especially for screening and in patients with corneal pathology,” Dr. Shazly said.
Dr. Chynn said that the device might be a good instrument for family practitioners who screen patients for glaucoma, because it does not require anesthesia and can be performed simply in an office setting.
“The [device] is easy to use, user friendly, and it can be used on patients for mass screening,” Dr. Shazly said. “It’s portable, it’s economical; it’s really good for screening purposes.”
Retrospective chart review
Richard S. Davidson, MD, of the Rocky Mountain Lions Eye Institute, Aurora, CO, led a retrospective char t review of consecutive IOP measurements per formed on 64 eyes of 32 patients aged 34 to 91 years with both tonometers. Dr. Davidson found that 83% of all measurements were within 2 mm Hg of each other.
“The transpalpebral method of measuring IOP with the (proprietary) tonometer correlates well with Goldmann applanation,” the study concluded. “(It) may be a clinically useful device for measuring IOP in routine eye exams.”
A similar retrospective review, with Theodore H. Curtis, MD, affiliated with the Rocky Mountains Lions Eye Institute at the time of the study and now in private practice near Portland, OR, found that the tonometer’s pressure measurements correlated well with measurements by another proprietary applanation tonometer (Tono-Pen, Reichert). The study found that the aforementioned device was useful when examining children who “were reassured by the fact that no drops were needed.”
The proprietary tonometer requires no anesthesia and can be performed in an office setting
John Hope, MD, an ophthalmologist in private practice in Oklahoma City, said that he prefers the device because applanation tonometry is time-consuming and often requires support staff. Dr. Hope said that he has used the instrument routinely on every patient for at least 6 months.
“This instrument is so user friendly . . . there are no rubber covers to deal with, and, after the initial purchase, it is virtually maintenance-free,” he said.“I clean the instrument with an alcohol pad after each use, and it is easily portable in your pocket and can be transported from room to room or office to office.”
“There is no corneal contact and pressures can be obtained in patients wearing contact lenses,” Dr. Hope concluded. “The technique is easily and quickly learned.”
fyi
John Hope, MD
Dr. Hope has no financial interest in the subject matter.
Mark Latina, MD
Dr. Latina has no financial interest in the products described in the article.
Take Home Message
A proprietary tonometer (Diaton, DevelopAll Inc.,) has been found to be comparable with the Goldmann tonometer in several clinical trials.
More details about Diaton Tonometer at www.TonometerDiaton.com
DevelopAll Inc,. 1-877-342-DIATONS (877-342-8667)