Unique Non-Corneal Diaton Tonometer to be Presented at ASCRS & ASOA Symposium & Congress
Diaton Non-Corneal Tonometer will be seen by over 7000 attendees of American Society of Cataract and Refractive Surgery and the American Society of Ophthalmic Administrators ASCRS/ASOA Symposium & Congress in Boston, Massachusetts.
World Cornea Congress VI & ASCRS Glaucoma Day will also be held.
Diaton tonometer presentations and first hand training will be offered at BiCOM Inc., Booth # 954 in the Boston Convention & Exhibition Center.
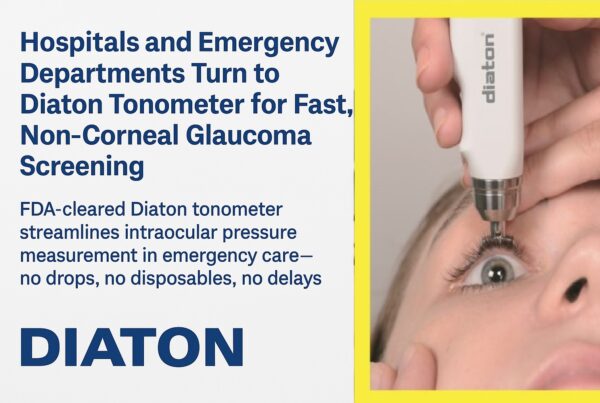
Diaton Non-Corneal and Transpalpebral tonometer presents unique ability of obtaining intraocular pressure (IOP) over the upper eyelid and over the sclera, independent of the corneal properties.
This non-invasive and non-corneal tonometry method enables to measure IOP in scenarios where otherwise all other corneal tonometers simply can not be used.
Roman Iospa, CEO of BiCOM Inc., said, – "Diaton non-corneal tonometer had finally solved the problem of the doctors and patients, where the patient was left not tested due to the corneal complications such as infection, patients sensitivity or some other reason. I'm glad my company is able to introduce a solution with this new non-corneal technology."
Diaton Tonometer is intended for use by Inpatient & Outpatient Clinics such as Hospitals, Emergency Rooms, Nursing & Elderly Homes, General & Specialty Practitioners as well as Ophthalmologists and Optometrists.
Tonometer Diaton is the solution in the following cases when the use of other devices is problematic or impossible:
* mass prophylactic screening of patients;
* IOP control during clinical observation of glaucoma patients (diurnal curve) ;
* ortoclinostatical probe, as an additional test to diagnose glaucoma and during select the adequate hypotensive therapy;
* ophthalmotone monitoring (even at night time);
* IOP measuring during contact correction (contact lenses are not taken out),
* IOP measuring in immobilized patients;
* IOP measuring in children (measurement is done outside of the visual field).
* on patients with the following conditions: chronic conjunctivitis, cornea pathology, including keratitis, keratotone, cornea dimness, after penetrating keratoplastics, keratoprosthesis, laser refractive correction of the eyesight, high degree of ametropy, astigmatism and others;
* on patients with medicinal allergies;
* Lasik / LASEK / PRK (recent clinical trials have proved that non-corneal Diaton is the only device that can be used for IOP measurement right after these surgeries)
More information on Diaton tonometer, including training/ Demo Video and clinical trials data can be found at http://www.TonometerDiaton.com or by calling BiCOM Inc. at the toll free number 1-877-diatons 1-877-342-8667.
Diaton tonometer is delivered complete with battery, training Video/DVD describing full procedure and tonometer cleaning instructions step-by-step in addition to the Test/Training Eye which is included with each unit.
About BiCOM Inc.
BiCOM, with its 18 years of tradition and global clientele, has proven to be the enterprise of the highest level of professionalism, integrity and financial solvency. A unique team of engineers, medical, legal and business experts makes BiCOM Inc. the right place for global talent to find support and guidance. BiCOM Inc. sees its mission in bringing to market innovative products developed and manufactured worldwide.
Diaton Tonometer is currently represented and has tonometer distributors in over 50 countries and growing. More about BiCOM or obtain information about distributors and distributorship in your country visit: http://www.TonometerDiaton.com
Press Contact –
Alina Lagoviyer / Media Relations
BiCOM Inc.
151 East Walnut Street
Long Beach, New York 11561
Phone: 1-877-342-8667
Fax: 1-888-260-0606
Contact@TonometerDiaton.com
http://www.TonometerDiaton.com